-
In order to regulate safety assessment and licensing of new food-related product varieties, the Center of Inspection and Supervision, Ministry of Health (which is reformed into National Health and Family Planning Commission now) formulated the “Regulations on Administrative Licensing of New Food-related Product Varieties”.
New food-related product varieties, refer to food packaging materials, containers, detergents, disinfectants, and new materials, new additives for tools and equipment in food production, including:
1.Food packaging materials, containers and additives which have not been included in the national food safety standards or approved for use in Ministry announcement;
2.Expansion of scope or amount in use of food packaging materials, containers and additives;
3. New materials which have not been included in the list of raw materials of food related disinfectant and detergent;
4. New materials and additives of tools and equipments, which are in direct contact with food in food production. -
Dossier for Submission
(A)The application form;
(B)The physical and chemical properties;
(C)Technical necessity, purpose and conditions of use;
(D)The manufacturing process;
(E)Quality specifications, test methods and inspection reports;
(F)Toxicological safety assessment information;
(G)Migration and / or residual amount, estimated dietary exposure and its assessment;
(H)Information or certified documents for the using approval of the materials in China or foreign countries;
(I)Other information favored for the assessment.Article (G) could be waived in application concerning food disinfectant and detergent.
Using scope, amount and other related information should be submitted in application concerning new additives in food packaging materials, containers, tools, and equipments.
Article (A), (C), (F), (G), and using scope, amount and other related information should be submitted in application concerning expansion of scope or amount of the additive in food packaging materials, containers, tools, and equipments.In application concerning first imported new varieties of food-related products, the following documents in addition to Article (E) should be submitted as well:
(A)Certified documents for the producing and selling approval issued by Authority of the country (or region), from which the products are exported;
(B)Producer certificate issued by Authority or Organization of the country, in which the products are produced;
(C)Commission contract for application submission;
(D)Validated Chinese translation of above documents. -
Declaration process
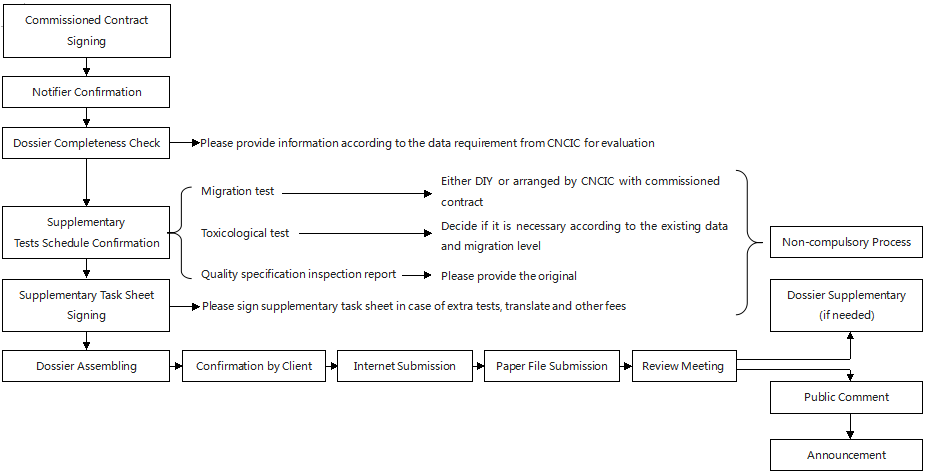
-
Our Services
Regulations consulting
Food safety regulations scope
Compliance assessment and analysis
Dossier preparation
Dossier review and professional advice for submission
Test arrangement
Review process tracking
Periodically reporting for progress







